Ocular Irritection® OECD 496 - Eye Irritation Test
Home » InVitro International » Eye Irritation Test OECD 496
The first and only 100% animal free eye irritation test adopted by the OECD
Ocular Irritection® OECD 496, the in vitro macromolecular test for the identification and classification of chemicals that inducesevere eye damage (UN GHS Category 1) and chemicals that do not require classification (UN GHS No Category).
- Globally accepted and recognized
- 100% animal free
- It can be carried out in 3 hours
- Results in 24 hours
- Easy to use

First and only OECD Test Guideline 100% animal free
TG OECD 492 uses a RhCE tissue model based on a human cell culture.
TG OECD 491 uses a rabbit corneal cell line.
TG OECD 437 and 438 make use of fabrics from abattoirs.
TG OECD 460 makes use of kidney cells.
Only Ocular Irritection® OECD 496 makes use of a fully acellular macromolecular Assay System.
Recommended by the OECD as a first test for both Bottom-Up and Top-Down strategy
Ocular Irritection® is recommended as an initial step in a Top-Down testing strategy, as described in the guidance document OECD 263, to positively identify chemicals that induce severe eye damage, i.e., chemicals to be classified as category 1 of the United Nations GHS without further tests.
Ocular Irritection® is also recommended to identify chemicals that do not require classification for eye irritation or serious eye damage as defined by the UN GHS (UN GHS No Category), and can therefore be used as an initial step within a Bottom-Up testing strategy approach.
Compatible with all plate readers
Tests compatible with any plate reader with 405 nm and 450 nm filters.
The test
Ocular Irritection® is a standardized, quantitative in vitro test that predicts the ocular irritation potential of surfactant and non-surfactant chemical, petrochemical, consumer, and cosmeticproducts, mixtures, or formulations.
Part 1
Part 2
How does it work?
The test is based on the understanding that the ability of a chemical to irritate the cornea is related to its propensity to promote denaturation and destruction of corneal proteins. Ocular Irritection® mimics these biochemical phenomena using a protein reagent.
When the assay is performed, the changes in protein structure induced by the test sample are then quantified by measuring the resulting changes in turbidity (optical density) of the solution reagent at a wavelength of 405 nm using a spectrometer.
The results of past comparative studies were used to develop what is called an “Irritection Draize Equivalent” (IDE) score, which is a value derived from in vitro studies that is equivalent to the expected irritation potential in vivo (Draize rabbit eye test).
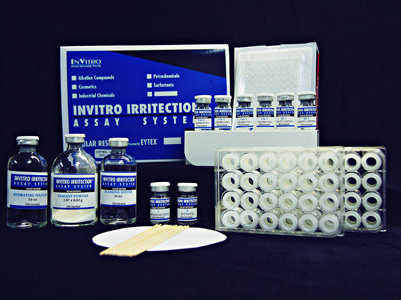
Ocular Irritection® is part of the Irritection® Assay System with Irritection® Dermal.
Ocular Irritection® is OECD TG 496, the first and only OECD in vitro test that is totally animal free.
According to the 11th revision of the SCCS concerning the “Notes of Guidance for the Testing of Cosmetic Ingredients and the Evaluation of their Safety“, at paragraph 3–4.6, Ocular Irritection® is the first and only OECD adopted test that is completely animal free.
Classifies both UN GHS Category 1 and UN GHS No Category, mixtures and formulations, surfactants and non-surfactants, solids and liquids
TG OECD 492 can only classify UN GHS No Cat.: The test must always be part of a bottom up strategy and cannot be used to classify UN GHS Cat. 1 and 2 substances.
TG OECD 491 is not applicable on solid chemicals other than surfactants and mixtures composed only of surfactants.
Do you perform ophthalmological testing? Do a preliminary screening with Ocular Irritection®.
The second general principle of the World Medical Association’s Declaration of Helsinki, updated 2013, on “Ethical Principles for Biomedical Research Involving Human Beings,” capstone reads:
“It is the physician’s duty to promote and safeguard the health, well-being, and rights of patients, including those involved in biomedical research. Physician science and co-science must be placed in service of this mission.“
The next point further states:
“Medical advances are based on research, which ultimately must be based on clinical trials conducted in humans.”
Don’t use your informed volunteer as a “guinea pig”, do a pre-screening ophthalmology test with Ocular Irritection®.

Irritection® Dermal
Dermal Irritection® is the technology that has accompanied Ocular Irritection® for over 30 years and provides GHS-accepted results through the weight of evidence.
Although they are two different methods, both make use of the Irritection® Assay System and employ matrices specifically designed to mimic the biochemical phenomenon of alteration of the proteins of the dermis or cornea when brought into contact with the substance under examination.


